For most people, metal is another word for iron, steel, or a similar hard, shiny substance.
But does this definition fit with the true properties of metals?
Yes… and no.
Before we explain, you should know that most of the elements in the periodic table are metals.
Metals are found in the center and left side of the periodic table. They can be further classified as alkali metals, alkaline earth metals, transition metals, and basic metals.
Metals Science Lesson
Properties of Metals
An element is a substance made up of one kind of atom; it cannot be separated into simpler parts. For example, the element helium (think hot-air balloons) is made up exclusively of helium atoms.
Elements are generally classified as metals or nonmetals (although some elements have characteristics of both; these are called metalloids).
Three properties of metals are:
- Luster: Metals are shiny when cut, scratched, or polished.
- Malleability: Metals are strong but malleable, which means that they can be easily bent or shaped. For centuries, smiths have been able to shape metal objects by heating metal and pounding it with a hammer. If they tried this with nonmetals, the material would shatter! Most metals are also ductile, which means they can be drawn out to make wire.
- Conductivity: Metals are excellent conductors of electricity and heat. Because they are also ductile, they are ideal for electrical wiring. (You can test this using some household items. Keep reading to find out how!)
Additional Properties of Metals
High melting point: Most metals have high melting points and all except mercury are solid at room temperature.
Sonorous: Metals often make a ringing sound when hit.
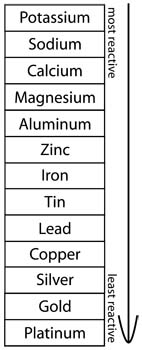
Reactivity: Some metals will undergo a chemical change (reaction), by themselves or with other elements, and release energy. These metals are never found in a pure form, and are difficult to separate from the minerals they are found in. Potassium and sodium are the most reactive metals. They react violently with air and water; potassium will ignite on contact with water!
Other metals don’t react at all with other metals. This means they can be found in a pure form (examples are gold and platinum). Because copper is relatively inexpensive and has a low reactivity, it’s useful for making pipes and wiring.
Five groups of metals:
Noble Metals are found as pure metals because they are nonreactive and don’t combine with other elements to form compounds. Because they are so nonreactive, they don’t corrode easily. This makes them ideal for jewelry and coins. Noble metals include copper, palladium, silver, platinum, and gold.
Alkali Metals are very reactive. They have low melting points and are soft enough to be cut with a knife. Potassium and sodium are two alkali metals.
Alkaline Earth Metals are found in compounds with many different minerals. They are less reactive than alkali metals, as well as harder, and have higher melting points. This group includes calcium, magnesium, and barium.
Transition Metals are what we usually think of when we think of metals. They are hard and shiny, strong, and easy to shape. They are used for many industrial purposes. This group includes iron, gold, silver, chromium, nickel, and copper, some of which are also noble metals.
Poor Metals are fairly soft, and most are not used very much by themselves. They become very useful when added to other substances, though. Poor metals include aluminum, gallium, tin, thallium, antimony, and bismuth.
Alloys: Strong Combinations
The properties of these different metals can be combined by mixing two or more of them together. The resulting substance is called an alloy. Some of our most useful building materials are actually alloys. Steel, for example, is a mixture of iron and small amounts of carbon and other elements; a combination that is both strong and easy to use. (Add chromium and you get stainless steel. Check your kitchen pots and pans to see how many are made from stainless steel!)
Other alloys like brass (copper and zinc) and bronze (copper and tin) are easy to shape and beautiful to look at. Bronze is also used frequently in ship-building because it is resistant to corrosion from sea water.
Titanium is much lighter and less dense than steel, but as strong; and although heavier than aluminum, it’s also twice as strong. It’s also very resistant to corrosion. All these factors make it an excellent alloy material. Titanium alloys are used in aircraft, ships, and spacecraft, as well as paints, bicycles, and even laptop computers!
Gold, as a pure metal, is so soft that it is always mixed with another metal (usually silver, copper, or zinc) when it’s made into jewelry. The purity of gold is measured in karats. The purest you can get in jewelry is 24 karats, which is about 99.7% pure gold. Gold can also be mixed with other metals to change its color; white gold, which is popular for jewelry, is an alloy of gold and platinum or palladium.
Metal from Ore
Ores are rocks or minerals from which a valuable substance – usually metal – can be extracted. Some common ores include galena (lead ore), bornite and malachite (copper), cinnabar (mercury), and bauxite (aluminum). The most common iron ores are magnetite and hematite (a rusty-colored mineral formed by iron and oxygen), which both contain about 70% iron.
There are several processes for refining iron from ore. The older process is to burn iron ore with charcoal (carbon) and oxygen provided by bellows. The carbon and oxygen, including the oxygen in the ore, combine and leave the iron. However, the iron does not get hot enough to melt completely and it contains silicates left over from the ore. It can be heated and hammered out to form wrought iron.
The more modern process uses a blast furnace to heat iron ore, limestone, and coke (a coal product, not the soft drink). The resulting reactions separate out the iron from the oxygen in the ore. This ‘pig iron’ needs to be further mixed to create wrought iron. It can also be used for another important purpose: when heated with carbon and other elements, it becomes a stronger metal called steel.
Considering the process involved, it’s not surprising that iron was not used until around 1500 BC. But some pure metals – gold, silver, and copper – were used before then, and the alloy bronze is thought to have been discovered by the Sumerians around 3500 BC. But aluminum, one of the most essential metals in modern use, wasn’t discovered until AD 1825, and wasn’t commonly used until the 20th century!
Corrosion: Process & Prevention
Have you ever seen a piece of silver that lost its shine, or iron with reddish-colored rust on it or even holes in it caused by corrosion? This happens when oxygen (usually from the air) reacts with a metal. Metals with a higher reactivity (such as magnesium, aluminum, iron, zinc, and tin) are much more prone to this kind of chemical destruction, or corrosion.

When oxygen reacts with a metal, it forms an oxide on the surface of the metal. In some metals, like aluminum, this is a good thing. The oxide provides a protective layer that keeps the metal from corroding further.
Iron and steel, on the other hand, have serious problems if they are not treated to prevent corrosion. The reddish oxide layer that forms on iron or steel when it reacts with oxygen is called rust. The rust layer continually flakes away, exposing more of the metal to corrosion until the metal is eventually eaten through.
One common way to protect iron is to coat it with special paint that keeps oxygen from reacting with the metal underneath the paint. Another method is galvanization: in this process, steel is coated with zinc. The oxygen, water molecules, and carbon dioxide in the air react with the zinc, forming a layer of zinc carbonate that protects from corrosion. Look around your house, yard, and garage for examples of corrosion as well as galvanization and other means of protecting metal from rust.
Technology: Fireworks & Chemistry
If you watch fireworks on the Fourth of July, you’ll see beautiful combinations of color and sparks.
How does this amazing pyrotechnics display work? The short answer is chemistry. The longer involves a recap of the properties of metals.
One of the key ingredients for firecrackers, ground fireworks, and aerial fireworks (ones which explode in the sky) is black powder, invented by the Chinese about 1000 years ago. It’s a blend of potassium nitrate (saltpeter), charcoal, and sulfur in a 75:15:10 ratio. Black powder is used to launch aerials and also causes the explosions necessary for special effects like noise or colored light.
In sparklers, black powder is mixed with metal powders and other chemical compounds in a form that will burn slowly, top to bottom. In simple firework rockets, black powder is confined in a tube around a fuse. When lit, the powder creates a force that results in an equal and opposite reaction, pushing the firework off the ground and then causing the compounds inside it to explode in the air.
More complex fireworks shells are launched from a mortar, a tube with black powder that causes a lift-off reaction when lit. The firework shell’s fuse is then lit as it goes up into the air, and at the right time an explosion inside the shell causes its special effects charges to burst.

The bright, colorful part of the fireworks display is caused by “excited” electrons in the atoms of different metal and salt compounds. These compounds are in little balls called stars, made of a similar compound to what makes a sparkler work.
Metals as Coloring Agents
Different metals burn in different colors; for example, if a copper compound is lit, its flame will be a blue-green color. Calcium burns red-colored and potassium burns purple. In fireworks, metals are combined to create different colors.
When the star compounds inside a firework are heated, the excited atoms give off light energy. This light falls into two categories: incandescence and luminescence. Incandescence is light produced from heat: in fireworks, reactive metals like aluminum and magnesium cause a burst of very bright light when they get hot — sometimes at temperatures over 5000 ° F!
Compounds that are less reactive don’t get as hot, resulting in dimmer sparks. Luminescence, on the other hand, is produced from other sources and can occur even at cold temperatures. The electrons in the compound absorb energy, making them “excited.” The electrons can’t maintain this high level, though, so they jump back to a lower level, releasing light energy (photons) in the process.
Barium chloride is a chemical compound that gives fireworks a luminescent green color, and copper chloride makes a blue color. For either kind of light, it’s important to use pure ingredients since traces of other compounds will obscure the color.





