 Science magic tricks, secret writing, petri dish snacks—throwing a science party is a perfect blend of the “wow factor” and hands-on fun.
Science magic tricks, secret writing, petri dish snacks—throwing a science party is a perfect blend of the “wow factor” and hands-on fun.
As you plan a science party, choose some projects to present as demonstrations and others that everyone can do.
Have an adult or older child pretend to be a “mad scientist” and do the demonstrations. Ordering lab coats and other authentic science supplies lend credibility and serve as fun science party favors and decorations.
Science Party Topics
Mad Scientist Demonstrations
Science Activities
Science Party Menu
Science Party Decor
Science Party Favors
Plan a Science Party:
Mad Scientist Demonstrations
The following projects are amazing to watch, whether at a science party or not!
Add your own dramatic flare to your demonstration and you’ll have a captive audience!
These projects require adult supervision.
Collapsing Cup
This project’s disappearing act will make guests wonder if they’re at a magic show!
Use chemistry to make a Styrofoam cup disappear before their eyes.
You can find acetone at a hardware store, or buy a small bottle of acetone here from us.
Acetone is very flammable, so keep it away from flames and use only in a well-ventilated area.
Watch the video, then try it yourself!
What You Need:
- Small aluminum pie tin or a glass bowl
- Styrofoam cup
- Acetone
- Safety gloves
- Safety goggles
What You Do:
1. Set the cup in the middle of the pie tin.
2. Carefully pour about a tablespoon of acetone into the cup.
3. Watch the reaction!
What Happened:
Styrofoam is mostly air, trapped in place by the polymer polystyrene.
A polymer is a very large molecule formed by repeated patterns of chemical units strung together in long chains.
The acetone dissolves the long polymer strands in the Styrofoam, allowing all the trapped air to escape.
Without air, the cup is just a blob of polystyrene on the bottom of the dish!
You can pour off the excess acetone and let the blob of polystyrene dry into a hard lump of plastic.
Mystery Pitcher
There’s a lot of opportunity to really ham it up when you do this demonstration!
Put on a show of being mysterious by showing the empty pitcher and having a guest fill it with water to prove you haven’t added anything to it.
Then watch them say “wow” when you change the water into “pink lemonade!”
(Make sure no one tries drinking any, though!)
Watch this project video for a preview. The video doesn’t use an opaque pitcher, but you’ll want to for your party.
What You Need:
- Phenolphthalein solution
- Sodium carbonate
- Vinegar
- 5 glasses and a non-see-through pitcher of water
What You Do:
1. Before your guests arrive, prepare the glasses. In the first glass put a little less than 1/8 teaspoon of sodium carbonate, in the second put 6 drops of phenolphthalein solution, and in the third put three dropper-fulls of vinegar.
2. Add a few drops of water to the first glass and stir to dissolve the sodium carbonate.
3. When it’s time to do the demonstration, fill all the glasses with water from the pitcher, then pour all of them back in the pitcher except for the glass with vinegar.
4. Refill the remaining four glasses – the water will be hot pink!
5. Now pour all five glasses back in the pitcher. Refill the glasses one last time—the liquid will be colorless again!
What Happened:
Phenolphthalein is a pH indicator that turns colors in reaction to bases.
When you poured the four glasses back into the pitcher, the phenolphthalein reacted to the sodium carbonate, a base, and turned the solution to pink “lemonade.”
To change it back to “water,” all you had to do was add the acidic vinegar, which lowered the pH and turned the phenolphthalein colorless again.
Plan a Science Party:
Hands-on Activities for Guests
Why should only one mad scientist have all the fun? Let everyone in on the fun with these science party activity ideas.
Spread out a disposable plastic tablecloth, pass out some lab aprons, and let your guests get to work!
Here are a few fun science party projects for party-goers:
Magic Writing
Everybody loves secret messages!
With adult supervision, let party guests write a note for another guest on white paper using a Q-tip dipped in phenolphthalein solution.
Once everyone has finished their message, pass the notes and let the recipient spray the paper with Windex.
The message will appear bright pink! Phenolphthalein is a pH indicator that turns pink in the presence of bases, like Windex.
Find other ways to write secret messages in our invisible inks project.
Water Gel Beads
 Let each guest get in on the mad scientist action with water gel marbles.
Let each guest get in on the mad scientist action with water gel marbles.
Pour some water gel beads into a cup of water, and watch them expand up to 400x in size!
The complete changes takes a few hours, so be sure to start the water jelly marbles soaking at the beginning of the party.
What happens if you add food coloring to the water first? Find out!
Plus, dried water beads in a baby soda bottles make a fun favor that will keep the fun going long after the science party is over.
Check out this water marbles project to see these superabsorbent polymers in action.
More Science Party Activities
You can also do STEM science projects that use only household items, like making slime or quicksand.
If you’re throwing your science party in summer, use the sun’s rays for fun STEAM projects, like making sunprints or stringing UV beads that change color in sunlight.
Making solar oven s’mores is another sunny weather idea.
During winter, make snow globes while learning about liquid density and play with instant snow powder.
Plan a Science Party:
Party Menu Ideas
Edible “Bacteria” Cultures
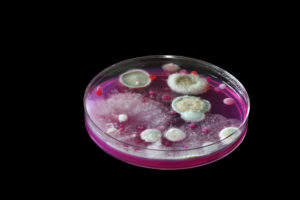
Petri dishes are used in a science lab to grow bacteria cultures on a gelatinous substance called agar. Mimic their scientific use by making jello and pouring it in clean petri dishes to set up. You can then use small candies like Nerds to create “bacteria colonies” on the surface of the jello.
Glow-In-The-Dark Jello
You can do this as a variation of the jello petri dishes, or make separate glowing “jigglers.”
To make glowing jello, use tonic water instead of regular water. Tonic water contains a chemical called quinine which fluoresces, or glows, under ultraviolet light.
When your jello is firm, shine a black light on it and watch it glow blue.
If you don’t like the taste of quinine, try using half tonic water and half tap water.
Dry Ice Punch
Add a new twist to your punch by adding some dry ice, available at grocery stores.
This looks very cool, as billows of “fog” will roll off your bubbling punch, and the dry ice will make the drink carbonated!
Dry ice is frozen carbon dioxide, and it is very cold—110 degrees below zero! This can cause severe thermal burns, so make sure you follow these guidelines:
1. Always have an adult handle dry ice. Use tongs and insulated gloves.
2. Make sure no piece of dry ice gets into someone’s glass. If possible, put one large block in the punch bowl, not lots of little pieces. Have an adult do the serving; you can serve it by pouring it through a strainer to catch any pieces of ice. (Unlike water ice, dry ice will sink, making it easier to avoid when serving.) You can also wait to serve it until all the dry ice has sublimated (changed from a solid to a gas).
Plan a Science Party:
Science Party Decorations
 Transform your venue into a lab with a few inexpensive science supplies.
Transform your venue into a lab with a few inexpensive science supplies.
Serve up food in an oversized beaker and pour drinks from an oversized flask.
An assortment of chemistry lab glassware makes attractive vases, too.
For older crowds, beaker mugs are a mad scientist party must!
Use a periodic table of the elements chart as a backdrop or prop.
Make your science party a bit creepy with preserved dissection specimens in jars or insect collections arranged in a display case.
Plan a Science Party:
Science Party Favors
Kids feel like real scientists with real science supplies! Lab coats let them dress the part at the science party. Giant test tubes can serve up snacks, and petri dishes are the perfect place to cultivate jello. What are some other ideas for science party favors? See if we have them, since many items at HST qualify for bulk pricing to save you money.
More Science Links
- Get even more ideas for a science party, including invitations and decorations, at About.com.
- Download this party-planning checklist to help you gather the supplies you’ll need.
- See another fun party demonstration with this egg-in-a-bottle project video.
- Explore science further with other fun chemistry sets from HST!





