When dissolved in water, acids donate hydrogen ions (H+). Hydrogen ions are hydrogen atoms that have lost an electron and now have just a proton, giving them a positive electrical charge. Bases, on the other hand, mixed with water yield hydroxide ions (OH-). If a solution has a high concentration of H+ ions, then it is acidic. If a solution has a high concentration of OH- ions, then it is basic.
In many acid-base reactions, the resulting product is water along and a salt. If hydrochloric acid (HCL) and the base sodium hydroxide (NaOH) are combined, the product is H2O (water) and NaCl (sodium chloride, a table salt). The H+ ions in the acid join with and are neutralized by the OH- ions of the base to form H2O.
One of the simplest activities to show how acids and bases react with each other (and to demonstrate their different properties) is to make a baking soda and vinegar ‘volcano’. To make a big eruption, use a small plastic bottle (the size 20-oz soft drinks come in works well). Fill the bottle halfway (1 to 1.5 cups) with vinegar. To start the eruption, drop a baking soda ‘bomb’ into the bottle—wrap one tablespoon of baking soda into a small piece of tissue paper, tying the ends with thread. You should see an instant eruption! The baking soda, a base, neutralizes the acid in vinegar. This releases carbon dioxide gas, which causes the fizzing action in your volcano. (An acidic solution is neutralized when a base is added to it, and a basic solution is neutralized by the addition of an acid.)
For another reaction experiment, put an Alka-Seltzer tablet in the bottom of a clear plastic film canister (the kind where the cap fits inside instead of closing over the outside). Fill the canister with warm water and then quickly put the cap on and watch the acid-base reaction!
The pH scale is used to measure the amount of H+ ions in a solution. Acids have a pH below 7; bases have a pH above. Strong acids have the lowest pH levels (0-4) and strong bases have the highest pH levels (10-14). Neutral solutions have a pH of 7 and they are neither acidic nor basic. Distilled water is neutral, because the H+ and OH- ions are balanced.
Litmus is a natural acid-base indicator extracted from a type of lichen. If you have red and blue litmus paper, you can test different solutions for whether they are acids or bases. Blue litmus paper turns red when a solution is acidic; red litmus paper turns blue in basic solutions. Try testing window cleaner, toilet bowl cleaner, orange juice, and apple juice—pour a little of each into separate test tubes or small glasses or jars. Use the litmus paper to determine which are acids and which are bases. Here are the pH levels of some other substances that you might test: lemon juice (2), vinegar (3), milk (6), egg whites (8), baking soda (9), and ammonia (10). Human blood has an ideal pH of 7.4; even slight fluctuations can seriously affect our bodies.
You can also make your own pH indicator—use a blender to mix one part chopped red cabbage with two parts boiling water and use the juice to test different solutions. Acids will turn the pigments in the indicator to a reddish color; bases will turn the pigments bluish or yellow-green.
Science Lesson: Good Water
Water may seem plain and boring sometimes, but it does have some interesting physical properties! Water weighs about 62 pounds per cubic foot at its freezing point; it freezes at 0 °Celsius and boils at 100 °; and it makes up about 60% of the composition of our bodies and 83% of our blood! As for what it can do, water is called the ‘universal solvent‘ because it can dissolve more substances than any other liquid, even the strongest acid.
The chemical composition of water is H2O—two hydrogen atoms and one oxygen. The way those atoms bond together to form a water molecule is what allows water’s special properties. The two H atoms form weak hydrogen bonds with the oxygen; they attach to the top of the molecule rather like Mickey Mouse ears.
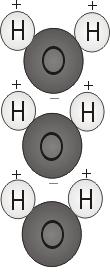
This molecular structure gives the water molecule polarity, or a lopsided electrical charge that attracts other atoms. The end of the molecule with the two H atoms is positively charged. The other end, with the oxygen, is negatively charged. Just like in a magnet, where north poles are attracted to south poles (opposites attract), the positive end of the water molecule will connect with the negative end of other molecules.
Water is the only natural substance that can exist in all three states of matter—solid, liquid, and gas—at the temperatures normally found on Earth. Many other substances have to be super-heated or -cooled to change states. The solid state of water (i.e., ice) is less dense than the liquid state, which is why ice floats.
Polar substances dissolve easily in water, the ‘universal solvent’. That is because one end of a polar molecule is more positively charged and one end is more negatively charged. The positively charged end of a polar molecule will be attracted to the negatively charged end of a water molecule, and vice versa. Likewise, positive ions are attracted to the negative end of the water molecule and vice versa. This molecular attraction is what allows so many substances to dissolve easily in water—the molecules are surrounded by water molecules. Oil and other non-polar solutions do not have positively or negatively charged ends, so water molecules will be attracted to each other rather than the oil. This is why oil does not mix with water.
Noteworthy Scientist: Sir Humphry Davy (1778-1829)
Davy was born in the county of Cornwall, England, on December 17, 1778. He was educated there, becoming apprenticed to a surgeon-apothecary when his father died. A few years later he moved to Bristol and became superintendent of a medical institution. At the age of 22, he wrote Researches, Chemical and Philosophical about his work on the effects of nitrous oxide (later known as laughing gas). He experimented with the gas, even inhaling it himself—one of the supposed causes of the sickness he suffered toward the end of his life.
The next work he did was in electrochemistry. His use of electrolysis (passing electric current through a substance to break it down into parts) led him to propose correctly that elements in a compound are held together by electrical forces.
Davy isolated the elements sodium, magnesium, potassium, calcium, boron (along with Gay-Lussac), and barium. He also determined that chlorine was an element rather than an oxygen compound, and he gave it its name (after the Greek word for ‘yellow-green’). He experimented with iodine, proved that diamonds are formed by carbon, and rightly suggested that acids contain hydrogen. Altogether, Davy isolated more elements than any other chemist did up till the 20th century.
Although he was known for his work in chemistry, Davy was more famous for his invention of a safe miner’s lamp. Methane gas in coal mines would explode on contact with the flame of the candles that the miners used for light, causing deaths and damage. Davy was asked to find a solution. He came up with a design for a lamp that surrounded the flame with fine wire gauze. Some light was still able to shine out, but the flame caused only contained explosions in the chamber where the gas entered the lamp; the gauze kept the heat of the flame from spreading to the outside gases. Davy’s method was used in later improvements of mine lighting.
During his lifetime, Davy was accorded numerous honors. He was elected a fellow to the Royal Society in 1803; two years later he received its prestigious Copley Medal; and in 1820 he was elected as its president. In addition, he received an award from Napoleon Bonaparte, who was impressed by his work in electrochemistry. He was knighted in 1812 under the Prince Regent (while George III was suffering from bouts of insanity) and received a baronetcy.





